Dr. Masafumi Ueda
Researcher in Organic Chemistry: Molecular Design toward Materials

Welcome to my website!
My name is Masafumi Ueda. I work as a university lecturer at Kitasato University, and specialize in Organic Chemistry.
This page is dedicated to the Ueda Group, a subgroup within the Advanced Organic Materials Lab, Department of Chemistry, School of Science, Kitasato University. We plan to update this page casually with introductions to our research, topics of our interest, and personal news.
Our Group focuses on the fields of "Structural Organic Chemistry" and "Organic Physical Chemistry", which examine the correlation between the molecular structure of organic compounds and their physical properties.
We conduct research together with students from our university (graduate and undergraduate students) to achieve the creation of novel molecular frameworks and the realization of unique physical properties.
Profile

Name Masafumi UEDA (University Lecturer, Ph.D)
Date of Birth 1 November 1988 (Ehime, Japan)
Address
Department of Chemistry, School of Science, Kitasato University
Advanced Organic Materials Chemistry Lab
1-15-1 Kitazato Minami-ku, Sagamihara, Kanagawa, Japan
Tel: +81-42-778-8158
Email: msfmueda[at]kitasato-u.ac.jp (Please transfer [at] to @)
Education
B.S., March 2011: School of Engineering, Ehime University (Prof. Y. Misaki)
M.S., March 2013: Graduate School of Science and Engineering, Ehime University
Ph.D., March 2016: Graduate School of Science and Engineering, Ehime University
Title: "Studies on Synthesis, Structures, and Properties of Novel [n]Radialenes Substituted with Multiple 1,3-Dithiol-2-ylidenes"
Research History
2013-2016: Part-time teacher, Shikoku Medical Technology College
2016-2017: Assistant Professor, School of Engineering, Hiroshima Univ. (Prof. A. Ikeda)
2017-2024: Assistant Professor, School of Science, Kitasato Univ. (Prof. Y. Mazaki)
2024-Current: Lecturer, School of Science, Kitasato Univ. (Prof. M. Hasegawa)
2025-Current: Visiting Researcher, The Institute of Organic Chemistry, the Polish Academy of Science (Prof. D. T. Gryko)
Researcher Numbers and IDs
KAKENHI Researcher # 60778611
ORCiD # 0000-0003-0378-7366
ResearchMap ID B000298815
J-GROBAL ID 201801007340630958

Our Research
Our group creates and designs novel π-conjugated molecules incorporating oxygen or sulfur atoms into hydrocarbon skeletons based on keywords such as "curved structures," "π-conjugated molecules," and "chalcogen elements."
We challenge ourselves to construct unique and beautiful molecular skeletons by leveraging organic synthesis techniques.
After elucidating the molecular structures, we investigate their unique physical properties, such as redox characteristics and luminescence properties, to clarify the correlation between molecular structure and physical properties, thereby advancing the development of molecular materials.
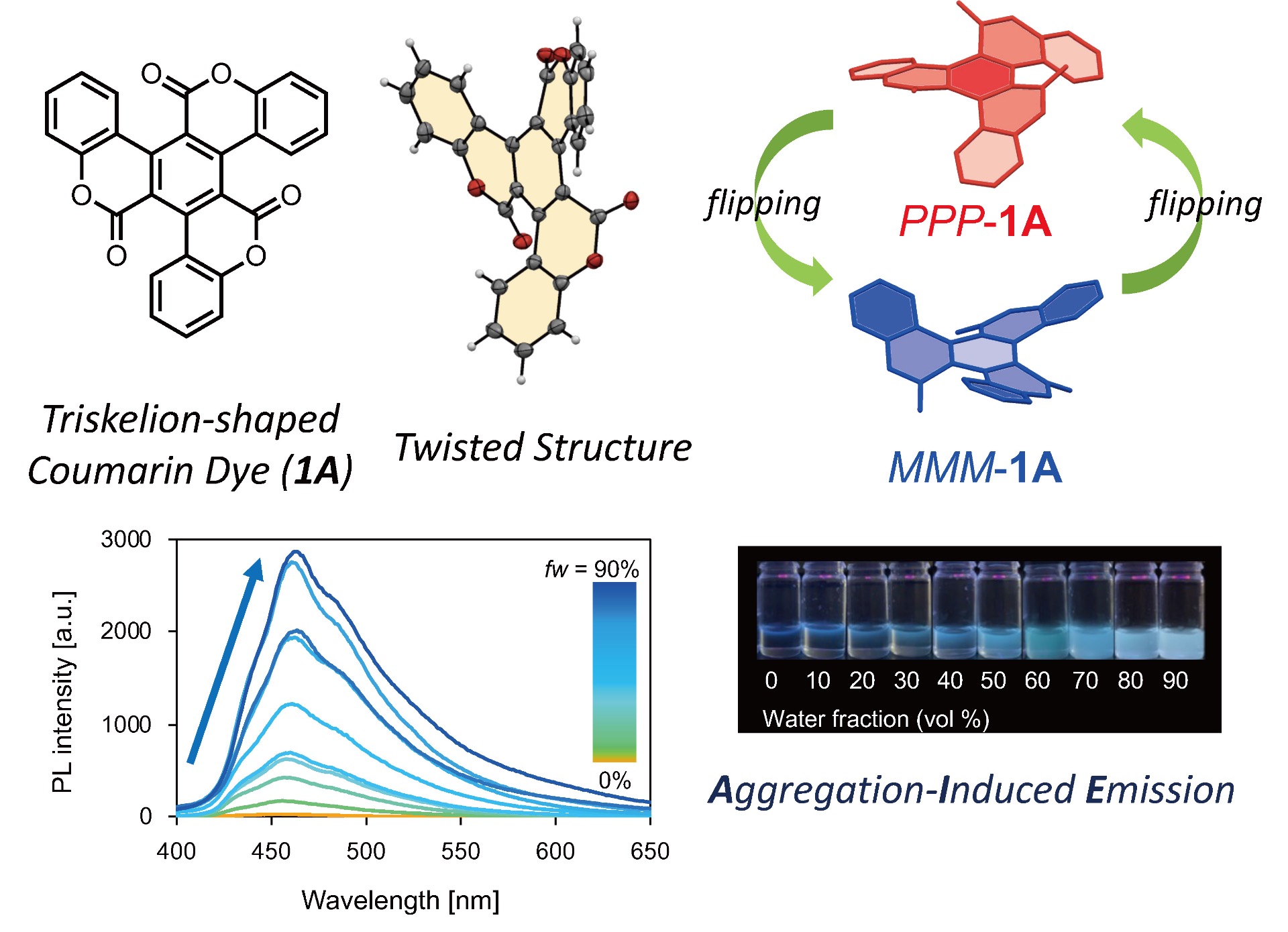
Development of Aggregation-Induced Emission Luminophores (AIEgens) Focusing on Molecular Inversion Behavior: Common π-conjugated dyes such as pyrene often adopt planar structures and exhibit highly efficient luminescence in dilute solutions. However, under high-concentration conditions, strong molecular overlap arising from the molecular skeleton leads to robust interactions causing stacking, resulting in concentration quenching. Conversely, sterically bulky compounds like hexaphenylsilole exhibit negligible luminescence in dilute solutions due to thermal deactivation caused by free rotation of phenyl groups. However, under high-concentration conditions, molecular aggregation suppresses the rotation of phenyl groups, leading to increased luminescence intensity. This phenomenon is termed "Aggregation-Induced Emission (AIE)", and compounds exhibiting this property are called "Aggregation-Induced Emission Luminogens (AIEgens)". These molecules, exhibiting a luminescence mechanism distinct from concentration quenching, have attracted significant attention from the perspectives of photochemistry and materials science. However, it is also true that many conventional AIEgens were synthesized based on designs that suppress "rotational motion" within the molecule, and the scope of application for AIE through molecular motion suppression remains undefined. Therefore, our group is focusing on π-conjugated organic dyes exhibiting "molecular inversion" behavior. By suppressing this inversion behavior, we are developing molecules that exhibit efficient luminescence in an aggregated state.
Selected Papers: ChemPhotChem (2020), BCSJ (2021), Eur. JOC (2024)
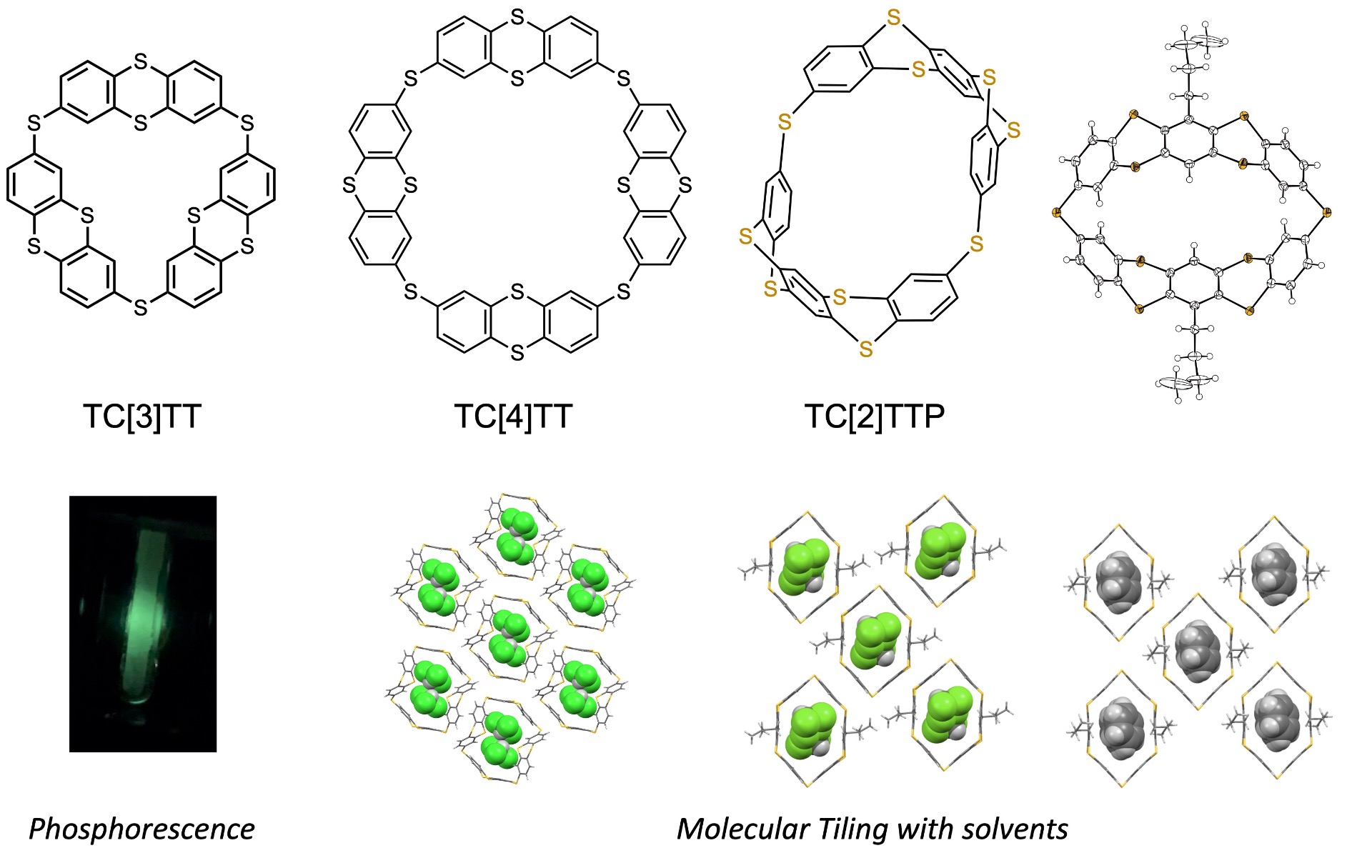
Development of Polygonal Macrocyclic Molecules Incorporating Sulfur-Containing Twisted Molecular Units: Macrocyclic molecules strongly attract our interest due to their beautiful radial molecular structures. Furthermore, possessing space dependent on the ring size within the molecule enables the incorporation of different organic molecules, potentially revealing novel properties based on molecular adsorption. Furthermore, molecules can be arranged in a columnar fashion along the ring structure, enabling the formation of compact molecular assemblies. This characteristic facilitates the evaluation of physical properties in the solid state. While the formation of the ring skeleton itself increases synthetic difficulty, typically resulting in low yields of the obtained compounds, it is anticipated that these molecules possess corresponding appeal and unique properties. Our group is developing polygonal macrocyclic molecules using folded polythiaacenes incorporating sulfur atoms—such as 1,4-dithiin, thianthrene, and tetrathiapentacene—as structural units, rather than generally planar aromatic π-conjugated molecules like benzene. Unlike cylindrical macrocycles, adopting polygonal structures enables the control of molecular arrangement based on mathematical "tessellation," termed "Molecular tiling." We are investigating how the photophysical and electrochemical properties of the building blocks influence the single-molecule and collective states when forming ring-shaped skeletons.
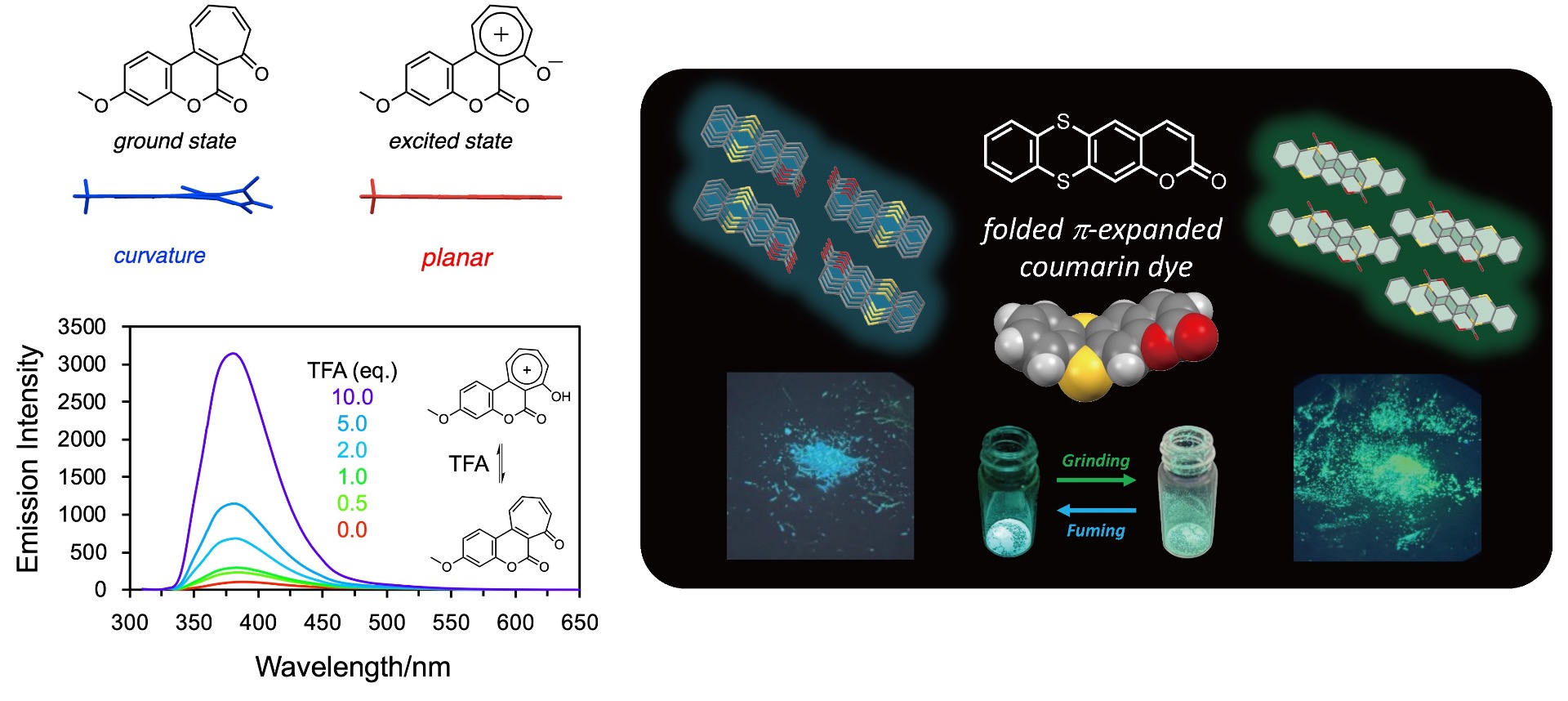
Development of External Stimulus-Responsive Luminescent Dyes: Organic luminescent dyes possessing a curved structure typically undergo a structural change from a curved to a planar structure when they absorb light and transition to an excited state. This process consumes energy through molecular motion. Consequently, during the decay process from the excited state (S1) to the ground state (S0) (S1→S0), "non-radiative deactivation"—where stored energy is released as heat—becomes dominant. This makes it difficult to develop organic dyes with high luminescence efficiency. However, if structural changes can be controlled by external stimuli such as mechanical pressure or pH changes, or by creating specific external environments through skeletal immobilization like aggregation or crystallization, these molecules can be developed as "switchable luminescent dyes" capable of turning luminescence on/off or dramatically increasing luminescence efficiency. Our group is developing conjugated luminescent materials that respond to pH or mechanical stimuli by chemically modifying coumarins and thianthrens, such as extending their π-conjugation.
Selected Papers: BCSJ (2022), Chem. Commun. (2026)
Development of Highly Efficient Organic Room-Temperature Phosphorescent Materials Based on the Thianthrene Skeleton: Thianthrene exhibits extremely low fluorescence emission efficiency (~2%) in solution due to structural changes occurring between the S1 and S0 states. However, when the molecular skeleton is immobilized within single crystals or thin films, it emits phosphorescence at room temperature, with its emission efficiency increasing dramatically (~25%). This is believed to occur because the immobilization of the molecular skeleton within the crystalline environment induces efficient intersystem crossing from the singlet excited state (S1) to the triplet excited state (T1) via spin-orbit coupling (SOC). Typically, phosphorescence is prone to reacting with ambient oxygen and is only observable at cryogenic temperatures (liquid nitrogen temperature), limiting its applications to specific environments. However, the ability to achieve phosphorescence at room temperature significantly expands its potential applications. Compounds exhibiting room-temperature phosphorescence are predominantly transition metal complexes, raising concerns about scarcity and environmental impact. Conversely, the development of room-temperature phosphorescent materials using organic compounds is expected to significantly contribute to materialization from the perspectives of resource availability, low environmental impact, and molecular design flexibility. However, the underlying emission mechanism remains largely unclear, and it is difficult to say that molecular design guidelines have been established. Our group is synthesizing derivatives based on thianthrene and developing materials that exhibit efficient room-temperature phosphorescence.
Selected Papers: In preparation


Project & Fundation
Japan Society for the Promotion of Science (KAKEN)
Project Principal Investigator
"Development of Organic Room Temperature Phosphorescent Material Utilizing Folding-Based Structural Characteristics"
Grant-in-Aid for Scientific Research (C) 2024-Current
"Creation of chiral space based on molecular arrangement control using Janus-type fluorophores"
Grant-in-Aid for Early-Career Scientists 2021-2023
Co-Investigator
"Creation of redox-active molecular assembly by macrocycles based on thienoacenes"
Grant-in-Aid for Scientific Research (C) 2022-2024
(PPI: Prof. Y. Mazaki from Kitasato University)
"Non-adiabatic transition dynamics in aggregation-induced emission"
2022-2025 Grant-in-Aid for Scientific Research (C) 2022-2025
(PPI: Prof. N. Yamamoto from Chiba Institute of Technology)
Other
"Design of Boron-Bridged Organic Dyes for Efficient Red/NIR Emission"
Nakatani foundation Study Abroad Program 2026
"Development of Emissive Materials using π-Extended Coumarin Derivatives"
Grant-in-Aid for Early-Career Scientists at Kitasato University 2018-2019
"Control of Drug Delivery by Photo-responsible Molecules"
Grant-in-Aid for Exploratory Research at Hiroshima University 2016-2017

Publication
Please refer to the following links for the original research papers published by our group

Contact
Masafumi UEDA (Ph.D, Lecturer)
〒252-0373 1-15-1, Kitazato, Minami-ku, Sagamihara, Kanagawa, Japan
+81-42-778-8158
msfmueda[at]kitasato-u.ac.jp
Please transfer [at] to @